Calculate the standard cell potentials of galvanic cell in whiCHM the following reactions take p... - YouTube

equilibrium - Calculate the cathode electrode potential in this redox reaction - Chemistry Stack Exchange

SOLVED: Calculate the standard cell potential of the following cell at 25'C: Pb(s) Pb2+ (aq) || Cu? + (aq) Cu(s) Standard Electrode (Reduction) Potentials in Aqueous Solution at 258C Cathode (Reduction) Half-Reaction

The standard electrode potential for Daniel cell is `1.1V`. Calculate the standard Gibbs energy of - YouTube

Standard Reference Electrode Standard Hydrogen Electrode (SHE) SHE: Assigned V Can be anode or cathode Pt does not take part in reaction Difficult. - ppt download

Calculate the standard electrode potential of the Ni^(2+)//Ni electrode , if the cell potential ... - YouTube

Calculate the standard cell potential in (V) of the cell in which following reaction takes place: Fe^2 + (aq) + Ag^ + (aq) → Fe^3 + (aq) + Ag(s) Given that E^oAg^ + /
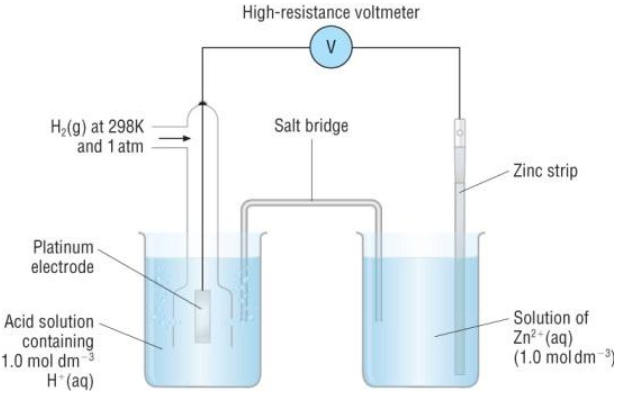
How to calculate the potential of zinc electrode capacity when in contact with 0.1M zinc sulphate solution in reference to hydrogen electrode when given the standard cell potential of Zn2 + /

The standard electrode potential (E^∘) for Daniel cell is + 1.1 V . Calculate Δ G^∘ for the reaction. Zn(s) + Cu(aq)^2 + → Zn(aq)^2 + + Cu(s) (1 F = 96500 C/mol)