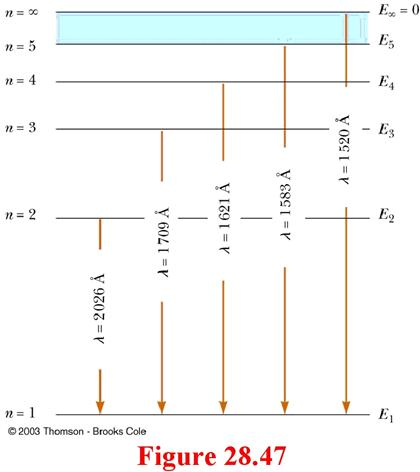
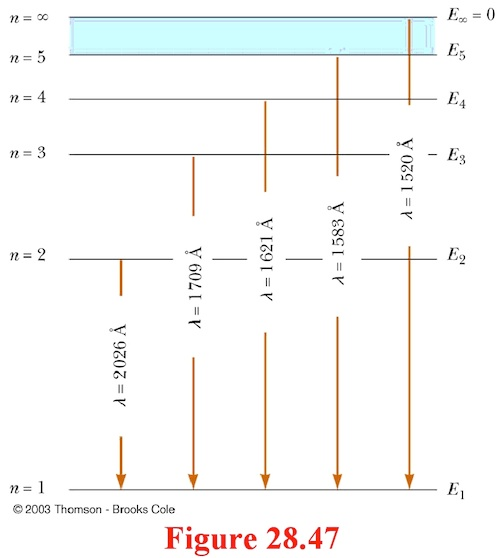
Using the Rydberg formula,calculate the wavelengths of first four spectral lines in the Lyman series of the hydrogen spectrum ? | EduRev Class 10 Question

The wavelength of the first line of Lyman series for hydrogen is identical to that of the second line of Balmer series for some hydrogen - like ion X . Calculate energies

The Lyman series for a one-electron atom is observed in a distant galaxy. The wavelengths of the first four lines and the short-wavelength limit of this Lyman series are given by the

SOLVED: The wavelengths in the hydrogen spectrum with m=1 form a series of spectral lines called the Lyman series. Calculate the wavelengths of the first four members of the series.

Stacked H I Lyman-series absorption-line profiles as a function of δ... | Download Scientific Diagram

Draw the energy level diagram of a hydrogen atom showing first four energy levels corresponding ton123and4 Show transitions responsible for absorption spectrum of Lyman series

Draw a Grotrian-type diagram for the first four lines of the Lyman series in the hydrogen atom spectrum. Be sure to include all allowed transitions. | Homework.Study.com

The four sets of lines in the hydrogen emission spectrum are known as Balmer, Brackett, Paschen, and Lyman. - Brainly.com